The Gods of Microbiology and the First Synthetic Drug
 |
| Louis Pasteur |
 |
| Robert Koch |
German
physician Koch was more willing to listen to his cohorts. He not only
isolated and identified the organisms responsible for Cholera,
Tuberculosis, and Anthrax, Koch inspired and instructed an entire
generation of German microbiologists who valued camaraderie.
Together, his pupils identify the causative organism and in many
cases effective treatments for typhoid, diptheria, gonorrhea,
leprosy, syphilis and many others. Koch made sure to give credit
where credit was due. Every hospital has a little room full of Petri
dishes, named for Koch s assistant Petri, full of agar medium which
was an idea of Koch's wife, upon which sputum and blood are streaked
in pursuit of marauding animalicules. Koch's postulates discredited
the Miasma theory of Disease, and the Germans could once again
gallivant confidently through the night air.
 |
| Paul Ehrlich |
In spite of Koch's non-competitive spirit, one can imagine the
trepidation of Paul Ehrlich when he found that both he was and Koch
were scheduled to speak on the same topic, methods of staining the
tuberculosis pathogen, at the Imperial Public Health Office. This
wariness likely grew to terror after Koch spoke, and Ehrlich realized
he, a mere student at the time, was going to stand before Koch and
his colleagues and explain the superiority of his method to Kochs.
Rather than chiding the upstart, Koch applauded Ehrlich and the two
began a life long friendship.
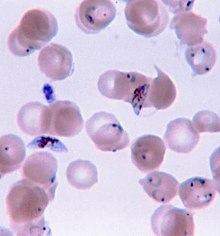 | |
| P. Falciparem |
While
Koch had effectively discredited the age old Miasma Theory of
Disease, those living near the equator still had reason to fear the
night air. Rather than Mal Aria or
“bad air,” the disease Malaria is spread by the bite of
mosquitoes which take flight at dusk. Unlike the majority of diseases
being studied during this seminal period of microbiology, Malaria
isn't caused by bacteria. The causative agent, Plasmodium Falciparem,
is a protist which is a type of single celled eukaryote. Eukaryotes
are millennia of milleninia ahead of bacteria in terms of complexity
and even today there are no effective vaccines against protists such
as those that cause Malaria, African Sleeping Sickness, and Giardia.
Ehrlich concluded that if a vaccine could not be produced to prevent
Malaria, then perhaps the agent itself could be attacked. He
theorized that because his favorite stain, Methylene Blue, binded so
readily to the Malaria pathogen, in higher doses it might even kill
it without harming the human host. He trialed methylene blue in two
Malaria patients; lo and behold, their fevers subsided and the nasty
little protists no longer showed in blood samples. This was the first
synthetic antibiotic agent, and it's discovery the herald of a whole
new means by which to treat the microscopic beasts within.
Bioenergetics,
Natto, and Whisky
Methylene
Blue (MB) has a rather unique means by which it kills organisms.
Understanding this mechanism requires a cursory understanding of
bioenergetics: the study of how energy flows through living systems.
It's important to become familiar with the compounds involved and a
great place to start is at the desired
end product: ATP.
|
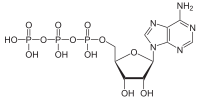
Adenosine Triphosphate (ATP)
All
known organisms use ATP to power protein machines within cells. It
consists of one Adenosine with three phosphate groups bound in a
sequence. When the high energy bond which links the third phosphate
is broken, energy is released in a way proteins can use, leaving behind an ADP and a phosphate.
|
There are many ways that ATP can be synthesized and bacteria are
particularly diverse in these pathways. Geobacter Metallireducens for
example uses uranium instead of oxygen as an electron receptor.
Archae breathe in carbon dioxide and breathe out methane gas. Amongst
Eukaryotes, yeasts are of particular note due to the inebriating
effects of the ethanol they produce. Ethanol does not result from the
ATP producing steps per se, but rather during fermentation which is a
way that the fungi can “reset” a very important intermediary
energy molecule called NAD.
|
Nicotinamide adenine dinucleotide (NAD)
NAD
is a coenzyme found in all living cells. It facilitates Redox
reactions in glycolysis and is reduced to NADH. NADH is then
transported to the mitochondria where it is oxidized back to NAD. This allows the NAD to once again facilitate redox reactions in glycolysis and provides the hydrogen and electrons which the Electron Transport Chain uses to synthesize ATP.
|
Production
of ATP starts outside of the body with a saccharide fuel source.
Regardless of whether this is healthy plump little grape or the last
extant twinkie, digestion begins in the mouth with salivary enzymes
that break down complex carbohydrates into simpler monomer forms such
as glucose. This happens two more times, once via enzymes from the
pancreas, and again from the brush borders of the small intestines.
Then,
absorption occurs. The glucose first enters the cells of the small
intestines. Then it enter the blood and travels around a bit before
finally being absorbed into the cell which will break it down for
energy. This is when “Cellular Respiration” begins, with the
process called Glycolysis. The
purpose of Glycolysis is to synthesize ATP the energy of life, but
the process isn't free.Glycolysis requires NAD.
 NAD
facilitates Redox reactions during glycolysis and are converted to
the reduced form NADH. This means that NAD accepts electrons in order
to bond with a hydrogen ion. This is essential for the payoff steps
of glycolysis to occur. Glycolysis is occurring pretty much
continuously in cells and organisms and must have a way to “reset”
the NADH back into it's oxidized form NAD. As discussed above, booze
producing yeasts such as Saccharomyces
cerevisiae use
a non-energy producing process called fermentation to reset NADH to
NAD. Fermentation is also used to make vinegar, pickles, and yogurt.
More exotic fermented foods include Surströmming,
a
swedish dish made of fermented Baltic Herring, and Natto, a fermented
soy bean dish from Japan with a texture often compared to snot.The
human body also performs fermentation if cells aren't receiving an
adequate supply of oxygen resulting in lactic acid. Accumulated
lactic acid is responsible for much of the muscle pain associated
with intense exercise. When sufficient amounts of oxygen are
available, the NADH is re-oxidzed instead by a series of enzymes
within the mitochondria, which make up the Electron Transport Chain.
NAD
facilitates Redox reactions during glycolysis and are converted to
the reduced form NADH. This means that NAD accepts electrons in order
to bond with a hydrogen ion. This is essential for the payoff steps
of glycolysis to occur. Glycolysis is occurring pretty much
continuously in cells and organisms and must have a way to “reset”
the NADH back into it's oxidized form NAD. As discussed above, booze
producing yeasts such as Saccharomyces
cerevisiae use
a non-energy producing process called fermentation to reset NADH to
NAD. Fermentation is also used to make vinegar, pickles, and yogurt.
More exotic fermented foods include Surströmming,
a
swedish dish made of fermented Baltic Herring, and Natto, a fermented
soy bean dish from Japan with a texture often compared to snot.The
human body also performs fermentation if cells aren't receiving an
adequate supply of oxygen resulting in lactic acid. Accumulated
lactic acid is responsible for much of the muscle pain associated
with intense exercise. When sufficient amounts of oxygen are
available, the NADH is re-oxidzed instead by a series of enzymes
within the mitochondria, which make up the Electron Transport Chain.
Redox
Reactions within The Electron Transport Chain
 Mitochondria
have an outer membrane and an inner membrane. The ETC is a set of
proteins complexes on the inner membrane which oxidizes NADH and
pumps the hydrogen ion (H+) into the space between the two membrane.
There, the H+ accumulates like air in a balloon, awaiting the
opportunity to escape. This concentration gradient of H+ is harnessed
by an enzyme called ATP-Synthase to make ATP. This is something like
a water-wheel in that H+ is allowed to flow, and it's movement powers
the ATP assembly mechanisms within the ATP-synthase enzyme.
Mitochondria
have an outer membrane and an inner membrane. The ETC is a set of
proteins complexes on the inner membrane which oxidizes NADH and
pumps the hydrogen ion (H+) into the space between the two membrane.
There, the H+ accumulates like air in a balloon, awaiting the
opportunity to escape. This concentration gradient of H+ is harnessed
by an enzyme called ATP-Synthase to make ATP. This is something like
a water-wheel in that H+ is allowed to flow, and it's movement powers
the ATP assembly mechanisms within the ATP-synthase enzyme. The
confounding nature of this terminology is evident when speaking about
compounds like NAD. NADH, albeit larger, is the reduced form of NAD
because NAD gains electrons in order to bind to the H+ and form NADH.
NAD is the oxidized form, in spite of the fact that the two compounds
have the exact same amount of oxygen. NAD is the oxidized form
because it lost electrons in order to kick out the H+. Complex I of
the Electron Transport Chain is responsible for this reaction.
Complex I strips away the electrons from NADH and pumps away the
hydrogen, leaving behind an NAD which can then be used again in
glycolysis.
The
confounding nature of this terminology is evident when speaking about
compounds like NAD. NADH, albeit larger, is the reduced form of NAD
because NAD gains electrons in order to bind to the H+ and form NADH.
NAD is the oxidized form, in spite of the fact that the two compounds
have the exact same amount of oxygen. NAD is the oxidized form
because it lost electrons in order to kick out the H+. Complex I of
the Electron Transport Chain is responsible for this reaction.
Complex I strips away the electrons from NADH and pumps away the
hydrogen, leaving behind an NAD which can then be used again in
glycolysis.
Methylene
Blue, like NAD, can pick up H+ and assume a reduced form. It's so
efficient at this process, that it can pull electrons out of the ETC
itself and stop the process. This is how Methylene Blue attacks
malaria. High doses of Methylene Blue inhibit the malarial
mitochondria from producing adequate ATP to continue living.
Methylene Blue essentially starves the malaria to death. The reason
it can be used safely in humans is that the ETC in malaria lacks a
few structures, such as complex I, which makes the malarial ETC more
susceptible to the theft of electrons by methylene blue.
Oxidative
Stress and The Palace of the Mad
In 1899, Paul Ehrlich assumed directorship of the Institute of
Experimental Therapy in Frankfurt near an insane asylum called, “Das
Irrenschloss...” The Palace of the Mad. It was here that Dr.
Alzheimer first described his eponymous disease which is first
expressed as a difficulty thinking abstractly and progresses to a
nearly complete loss of memory. Alzheimer Disease (AD) progresses
until sufferers can no longer perform even the most basic activities
to sustain life. During the 1980s, the Cholinergic Hypothesis
emerged, claiming AD results from a reduction in the ability to make
the neurotransmitter Acetylcholine. The majority of medications
prescribed for AD works to increase Acetylcholine. Unfortunately,
they've shown no impact on the progression of AD, and only moderately
decrease its symptoms. In the 1990s, research was focused on the
Amyloid Hypothesis. The brains of AD patients were found to contain
extensive deposits of Beta-Amyloid Plaques. These plaques were though
to be the underlying cause and an experimental vaccine was developed
to prevent plaque formation. This vaccine was successful, but in
spite of the absence of plaques, patient continued to grow
progressively worse and die. Further research on these beta-amyloids
revealed they have a non-pathological role and are found to a lesser
extent in all healthy adults.
Beta-amyloids
function to protect us against oxidative stress and their
overabundance in those suffering from AD seems to indicate that its
cause is associated with the most notorious electron leaking,
super-oxide producer in the cell: the Electron Transport Chain.
Research in the first decade of the twenty first century has shown
that those with AD exhibit a depression of all electron transport
chain complexes possibly due to oxidative damage. As these complexes
become less numerous and less efficient, more leakage of electrons
occur, and the ETC is further damaged. If one could find a substance
capable of picking up excess electrons which leak out of the ETC and
prevent the formation of superoxides, it would be possible to slow
the progression of AD; furthermore, if that same substance could then
return those electrons to the ETC, cells would have an increase in
available energy.
Methylene
Blue is this substance. The proposed trade name is “Rember.” A
2008 clinical trial of low dose Methylene Blue found an 81% reduction
in the speed of disease progression and trials are currently underway
to see if Methylene Blue is capable of preventing formation of AD
before symptoms arise. MB not only slows the disease, patients
demonstrate a significant improvement in memory function. It's not a
cure, but all research so far indicates that MB is the most promising
treatment for AD found so far. Research has also indicated that low
dose MB may be efficacious in the treatment of anxiety, depression,
and Parkinsons disease.
While
these finding are certainly exciting for those suffering from such
diseases, I most likely don't have Alzheimer's and I certainly don't
have Malaria. The paramount data yielded from research on methylene
blue is that in normal healthy adults it can improve memory, decrease
anxiety, and perhaps even slow the aging process. Rather than
interpreting the research for you, you'll find eight articles
addressing these three effects of interest.
IMPROVED
MEMORY
1. Behavioral, Physiological and Biochemical Hormetic Responses to the Autoxidizable Dye Methylene Blue
ANXIOLYTIC
DELAY
OF CELLULAR SENESCENCE
9.
Protective
Role of Methylene Blue in Alzheimer's Disease via Mitchondria and
Cytochrome c Oxidase
Dosage
Calculations
Take
a look at these values taken from the articles listed above:
50mg kg-1
|
Study 1
|
Amnesic Effects
|
5mg kg-1
|
Study 1
|
No effect noted
|
4mg kg-1
|
Study 2
|
Improved object memory recognition
|
1mg kg-1
|
Study 1
|
Enhanced memory retention
|
Study 3
|
Enhanced discriminative learning
|
|
0.05mg kg-1
|
Study 1
|
Enhanced memory retention, less than 1mg kg-1
|
It
would seem from these values that we want a dose higher than 0.05mg
kg-1,
but less than 4mg kg-1;
however, don't forget that these values are for rats! First we have
to do Allometric
Scaling, which is a kind of animal-to-animal dose conversion. The
underlying concepts are pretty interesting, but for brevity sake,
I'll spare you the details. After scaling, our values are as follows:
2.77mg kg-1
|
This dose is the human equivalent of 4mg kg-1,
and represents our upper limit
|
0.069mg kg-1
|
This dose is the human equivalent of 1mg kg-1
and represents our goal dose
|
0.035mg kg-1
|
This dose is the human equivalent of .05mg kg-1
and represents our lower limit
|
Now
that we have our human equivalents, we can move onto the next
problem... these rats were receiving MB intravenously. According to
the article, Pharmacokinetics
and organ distribution of intravenous and oral methylene blue,
only 60% of methylene Blue is absorbed through the GI tract. So we
have to correct for this:
4.61mg kg-1
|
2.77mg kg-1/ 0.6 = 4.61mg kg-1 is
the oral upper limit
|
0.115mg kg-1
|
0.069mg kg-1/0.6 = 0.115mg kg-1 is
the oral goal dose
|
.058mg kg-1
|
0.035mg kg-1/0.6 = .058mg kg-1 is
the oral lower limit
|
According
to these values, dosage should fall between 58 ug and 4.6mg per kg.
If ones isn't familiar with the metric system, you'd first divide
your weight in lbs by 2.2 and then multiply by 0.115mg. If I weigh
180 lbs, then my MB dose would be 9.4mg. This might seem a rather
reasonable number when one compares it to the 60mg proposed dose for
“Rember.”
Interestingly, the majority of experimental users of MB report the
greatest results from a dose three orders of magnitude lower. Many
people report the greatest benefits from a dose of 60 micrograms. That's a
mere 0.006 mg, yet many swear by it. Before trying MB for yourself, it would behoove to read some of the experiences reported by self-experimenter on forums such as Longecity
or Mind and Muscle.
Because of these vastly different
quantities, I'm going to provide two different dilutions for
Methylene Blue. The first method will provide a dilution where each
drop contains 60 mcg. I'd advise anyone using this augmentation to
start with the lowest dose and working up toward the larger doses
until one finds their own optimal amount. The second method will
provide 1mg per drop to assist those who choose to trial the higher
doses as used in the studies above. The nice thing about MB is it's
safety. According to NIH Toxicological studies, the LD50 for rats is
1250mg kg-1.
The average human doesn't show any indication of toxicity under
600mg, so playing around with doses between 60mcg and 10mg is pretty
harmless as long as your not on an SSRI and don't suffer from Favism.
Availability
Methylene
Blue is widely available, perfectly legal to own, and cheap as dirt.
It generally comes in one of two different concentrations.
The 1%
concentration is available from chemical and science supply houses
and is used as a biological stain and as an indicator in redox
reactions. A 1 ounce bottle goes for as little as $3.50 plus shipping.
I have purchased this type from The
Science Company
A 2.303% solutions can be bought from pet stores and aquarium supply
shops. It's an effective treatment for fungal infections and
protozoa such as Ich which afflicts fish. A 4 ounce bottle retails for around 8 dollars, so this is the most economic way to purchase MB.
A
third source is from suppliers such as Provepharm, which claim to
produce a better product with less heavy metals. The “cheap”
aquarium MB doesn't harm fish though... and fish are far more
sensitive to heavy metals than people. I'd advise you to save your
money and buy the cheaper product unless you have significant amounts
of disposable income.
DILUTION
To
accurately dilute your methylene blue, you'll need:
There
are guides which explain how to do these dilutions with an eye
dropper, but the volume from droppers varies by as much as 20%. It's
worth dropping 40 dollars to do it right, particularly since a single
bottle of methylene blue contains far more than a years worth of
doses. It isn't unusual to spend 50$ a month on a single Nootropic
such as Oxiracetam, so this is good investment.
The
dilution formula is:
V1 x C1 = V2 x C2
V1 x C1 = V2 x C2
V1 is the starting volume needed to make the solution
C1 is the concentration of starting solution
V2 is the volume of the final solution
C2
is the concentration of the final solution
It's
a simple formula to use and if one was feeling lazy they could
instead choose to use one of the many Online
Dilution Calculators. To be nice though, I'm going to walk you
through each math problem. (Thanks for the format Elus. It was more
visually appealing than anything else I could come up with.) All that's left is
for you to choose the dosage you want and use the formula for the
percent concentration of MB that you bought.
Diluting
a 2.303% solution to 60mcg/ml
This is the calculation to dilute 2.303% MB solution down to 60
micrograms/ml with a final volume of 100ml. Each ml of the final
solution will provide a 60mcg dose and there will be 100 doses
available.
V1
x (0.023 grams/mL) = (100mL) x (60 x 10-6 grams/mL)
...Solving for V1
we get
V1 = 0.26 mL
Therefore, we mix 0.26 mL of 2.303% MB solution and 99.74 mL water to
obtain a 60 micrograms per mL solution of MB with a total volume of
100 mL. This is how it's done:
- Using a 1mL pipette, draw up 0.26 mL of 2.303% MB solution
- Place MB solution in a 100ml graduated cylinder
- Fill the graduated cylinder to the 100ml mark with purified water
One mL of this solution is a 60 micrograms dose.
Diluting
a 1% solution to 60mcg/ml
This is the calculation to dilute 1% MB solution down to 60
micrograms/ml with a final volume of 100ml. Each ml of the final
solution will provide a 60mcg dose and there will be 100 doses
available.
V1
x (0.01 grams/mL) = (100mL) x (60 x 10-6 grams/mL)
...Solving for V1
we get
V1 = 0.6 mL
Therefore, we mix 0.6 mL of 1% MB solution and 99.4ml water to
obtain a 60 micrograms per mL solution of MB with a total volume of
100 mL. This is how it's done:
- Using a 1mL pipette, draw up 0.6 mL of 1% MB solution
- Place MB solution in a 100ml graduated cylinder
- Fill the graduated cylinder to the 100ml mark with purified water
One mL of this solution is a 60 micrograms dose.
Diluting
a 2.303% solution to 1mg/ml
This is the calculation to dilute 2.303% MB solution down to 1
milligram/ml with a final volume of 100ml. Each ml of the final
solution will provide a 1 mg dose and there will be 100 doses
available.
V1
x (0.023 grams/mL) = (100mL) x (1 x 10-3 grams/mL)
...Solving for V1
we get
V1 = 4.35 mL
Therefore, we mix 4.35 mL of 2.303% MB solution and 95.65 mL water to
obtain a 1 milligram per mL solution of MB with a total volume of 100
mL. This is how it's done:
- Using a 5mL pipette, draw up 4.35 mL of 2.303% MB solution
- Place MB solution in a 100ml graduated cylinder
- Fill the graduated cylinder to the 100ml mark with purified water
One mL of this solution is a 1 milligram dose.
Diluting
a 1% solution to 1mg /ml
This is the calculation to dilute 1% MB solution down to 1
milligram/ml with a final volume of 100ml. Each ml of the final
solution will provide a 1 mg dose and there will be 100 doses
available.
V1
x (0.01 grams/mL) = (100mL) x (1 x 10-3 grams/mL)
...Solving for V1
we get
V1 = 10 mL
Therefore, we mix 10 mL of 1% MB solution and 90.00 mL water to
obtain a 1milligram per mL solution of MB with a total volume of 100
mL. This is how it's done:
- Using a 10mL pipette, draw up 10 mL of 1% MB solution
- Place MB solution in a 100ml graduated cylinder
- Fill the graduated cylinder to the 100ml mark with purified water
One
mL of this solution is a 1 milligram dose.
Tips
and Technique
 Sometimes
people unfamiliar with lab equipment get confused by the numbering on
pipettes. They are labeled so that one measures as they eject the
fluid, so if you want 0.26 mL, you draw up an entire mL of solution
and then eject it out until you get to the 0.26 line. This wastes a
negligible 0.73 mL of MB, so if you want to be thrifty you can
subtract the amount you want (0.25 mL) from 1 mL. If you draw up to
the mark on the pipette showing the difference, in this case the 0.73
mark, you'll have exactly 0.26ml.
Sometimes
people unfamiliar with lab equipment get confused by the numbering on
pipettes. They are labeled so that one measures as they eject the
fluid, so if you want 0.26 mL, you draw up an entire mL of solution
and then eject it out until you get to the 0.26 line. This wastes a
negligible 0.73 mL of MB, so if you want to be thrifty you can
subtract the amount you want (0.25 mL) from 1 mL. If you draw up to
the mark on the pipette showing the difference, in this case the 0.73
mark, you'll have exactly 0.26ml.
Reading
the amount in a graduated cylinder is also a bit tricky if you have
no experience. You'll find that you don't get a nice flat line at the
top of the fluid. Instead, the fluid forms a meniscus with the sides
being higher and the center of the surface being lower. The volume is
always read from the lowest point of the meniscus.
As
I stated before, eye droppers are notoriously inaccurate but they are
more
convenient then carrying around a 1ml syringe. If you choose to use
an eye dropper, 20 drops usually equals around 1ml. One last tip:
Methylene Blue is a stain. Whatever it touches, be it your counter,
your carpet, your clothes or your skin... it is now blue. Skin is
constantly being shed, so stains on your hands will eventually fade.
Stains to dental work such as crowns and dentures will not. While the
low concentrations of MB aren't an issue, larger concentrations such
as the 1mg/ml might if held in the mouth for long. I've also read
about people using alternative means of administration such as
through a mucosal administration device, or via sublingual route. I
simply haven't seen research indicating these routes to be any better
than by mouth.
If
you read through forums, you'll also find reports of people combining
MB with 1-2 grams of vitamin C. This is rather cool to do because the
MB bonds to the excess hydrogen ions yielded by ascorbic acid, and
the solution turns clear. Some claim this somehow makes the MB work
more effectively, although I have my doubts and there isn't any
supporting research. Because the doses of MB we're discussing
maintain serum levels near homeostasis because the reduced and
oxidized form, the “starting state” of the MB should be
irrelevant. Vitamin C is good for you though! The color change is a
neat trick, and it makes it into a kind of sour-orange cocktail.
Precautions and Contraindications
Of
course, prior to using any substance, you should check with your
medical doctor. I'm not advising you take anything and you should do
your own due diligence. I'm simply reporting on something I find
interesting. Methylene blue has a number of other medical uses we
haven't discussed in the treatment of cyanide poisoning, an
interesting condition called Methemoglobinemia. A famous example of
people with methemoglobinemia are the Blue
Fugates of Troublesome Creek,
who were known for their vivid blue skin coloration. The condition is
a hereditary disorder of the blood which requires treatment with
oxygen and methylene blue. MB is administered at a dose of 2mg Kg-1
IV over as little as 5 minutes. Because we corrected for GI
absorption rates, this value is nearly the same dose as the animal
studies were using. Although adverse reactions aren't common, they do
occur. Side effects can include hypertension and chest pain.
Sometimes people experience confusion, dizziness, and headache. GI irritation can occur and both
the feces and urine can be stained blue but this isn't really
harmful. MB is damn safe under most circumstances but there are two
contraindication that if not heeded can be downright lethal:
Antidepressants and “favism.”
In high doses, MB
has been found to exhibit MAO-I properties. When an MAO-I is combined
with a serotonergic drug such as Prozac, Paxil, or Zoloft, an excess
of the neurotransmitter serotonin can cause a number of serious
psychiatric symptoms as well as CNS toxicity. Now, I've also read a
considerable number of subjective accounts claiming that low dose MB,
specifically the 60mcg dose, actually augments the action of these
antidepressants; there are obviously no studies that support this and
a serotonin storm isn't a good thing.
Favism is an
antiquated term for a hereditary disorder that results in a
haemolytic response to the consumption of broad beans, which are
called “fava” beans in Italy. The condition is now called a G6PD
deficiency. Methylene Blue is amongst a long list of drugs which can
cause hemolytic anemia in sufferers.
As with any
substance to which your body is naïve, start small. Some users have
even reported effects from doses as low as 5mcg. This rings of
crockery like homeopathy to me but if you find it works best for you,
my opinion is irrelevant. I haven't personally gone above a 2mg total
dose because it was less effective for me. I felt “cloudy” and
tired, thus I haven't even tried the dosing used in the animal
studies. Of course, curiosity will get the better of me sooner or
later... The vast majority of MB proponents really are using a mere
60mcg dose and so this is a great place to start.
Conclusion
Sources
http://www.ncbi.nlm.nih.gov/pubmed/17428524?ordinalpos=6&itool=EntrezSystem2.PEntrez.Pubmed.Pubmed_ResultsPanel.Pubmed_DefaultReportPanel.Pubmed_RVDocSum
Holmes
C. Long-term effects of Abeta42 immunisation in Alzheimer's disease:
follow-up of a randomised, placebo-controlled phase I trial. Lancet.
2008;372(9634):216–23. doi:10.1016/S0140-6736(08)61075-2.
PMID
18640458.





Thank you so much for going in details and explaining it so the average person can grasp the content. You actually made me really interested in biology and chemistry again =)
ReplyDeleteHi, this is a very informative article, but something troubles me about the calculations. You have considered total intestinal absorption of MB. However, GI doesn't absorb it all at once, but rather slowly releases it in the bloodstream, and it has been said that peak blood concentration of MB after an iv dose is 10 times greater than after an oral dose. So, do you think it should be accounted for and values for iv administration multiplied by 10 to give appropriate dose values for oral ingestion?
ReplyDeleteHave you found something regarding that?
DeleteHi, I really appreciated your dosage analysis and how to prepare the solution. I have been scouring the internet for pharmaceutical grade methylene blue, because I would feel more confident in the product if it wasn't intended for staining microscope slides or as a fish disinfectant. The chemical synthesis of methylene blue involves heavy metals and solvents like dioxane/carbon tetrachloride, so I feel that concerns about purity/contaminants are legitimate. The only sources of pharma grade methylene blue I could locate are a seller on ebay and this website: http://bluebrainboost.com. Do you have any idea whether these sources are legit?
ReplyDeleteHey anon, I have no idea; however, there's a great post here: http://www.brainprotips.com/quelling-synaptic-wear-and-tear-with-methylene-blue-dosage-safety-protocol-and-theoretical-underpinnings/.
ReplyDeleteThe advantage of this method is that the author is describing the use of a pure reagent being diluted rather than serial dilutions of an inferior source like I've described above. Good stuff! Check it out.
Another benefit of MB:
ReplyDeleteMethylene Blue Reverses Human Cell Aging, Treats Progeria Symptoms in New Study:
http://nutritionreview.org/2015/12/methylene-blue-reverses-human-cell-aging-treats-progeria-symptoms-in-new-study/
My uncle once suffered from Alzheimer's disease,i never thought there is a cure to it, we have used different medication but no solution,
ReplyDeleteI got a herbal product from Dr Benard for the first two month my Uncle took the medication, he was about to remember past,he was also getting back his memory. now my uncle is living a normal life,no more sign of Alzheimer. he is completely cured. any one with such case can contact Dr Phillip on benardleo13@gmail.com
If your goal is blue schlera then why not put a very small dose in your eyes? It stains your tongue so I'm sure it would stain your eyes. I'm a dune fan too so this is a tempting experiment. I wonder what dilution would be required to not cause damage. Looks like someone else had it in their eyes and it wasn't good. http://www.ncbi.nlm.nih.gov/pubmed/16670488 Maybe if you used a very very very small amount and then slowly increased the dose until you reach the desired eye color.
ReplyDeleteWhat does the negative exponent mean in the dosage ranges "less than 1mg kg-1" ? I haven't seen anything expressed this way before. Thanks.
ReplyDeleteThat's a standard scientific usage denoting "per." "1mg kg-1" is the same as saying "1mg per kg."
DeleteAppreciatte your blog post
ReplyDelete